By Bryan Spann, Fort Meade Public Affairs Office

Tuesday, General Paul Nakasone, Commander USCYBERCOM, Director National Security Agency/Central Security Service gets briefed ahead of his Covid-19 vaccination by Fort Meade MEDDAC Commander, Colonel Tracy Michael.
Fort Meade starts COVID vaccinations
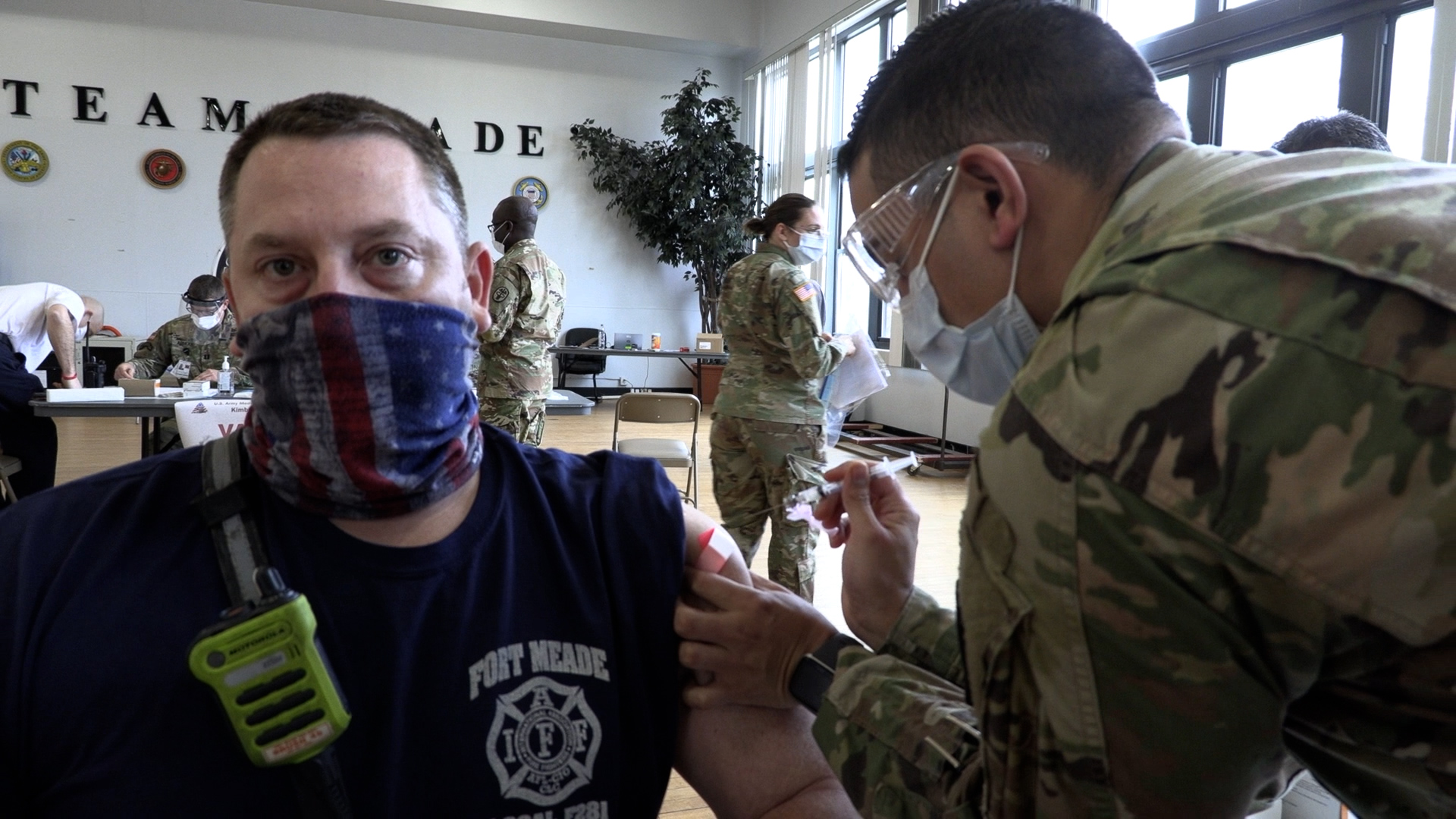
Kimbrough Ambulatory Care Center received an initial shipment of the Moderna COVID-19 vaccine just before the New Year. On Monday, the first 10 doses were administered to Fort Meade Fire and Emergency Services personnel at the McGill Training Center.
“We're hoping that in the first two to three weeks to get through the phase 1a category that includes healthcare workers and first responders on the installation,” says Col. Kimberly Salazar, Deputy Commander for Clinical Services for the Fort Meade Medical Department Activity.
The Moderna product is a two-dose vaccine with the doses taken 28 days apart.
“The exciting news from the clinical trials is…it's over 94 percent effective,” said Salazar. “So, it's a really important tool in this pandemic in terms of being able to, at some point, return to normal living.” She also added that getting the vaccination is voluntary, and acknowledges that people may have a lot of questions about the vaccine.
“One of the things that, in our planning phase, we really wanted to focus on was making sure we had an efficient and safe process for all of the beneficiaries coming in to receive the vaccine. It's a new vaccine, so we have a provider on site to answer any questions that beneficiaries may have before they receive the vaccine. It's completely voluntary, so they can opt out and choose not to receive the vaccine after they receive all the information,” she says.
Safety protocols at the immunization site include a monitoring area. All recipients of the vaccine are required to wait 15 minutes after receiving the vaccine in case signs of an allergic reaction appear. “We have all the supplies and medications here on site to treat a reaction if it were to occur,” says the colonel, “If they have a higher risk for an allergic reaction, we might ask them to wait longer, up to 30 minutes. So we try to think through all those potential scenarios. It's always good to plan for what could go wrong. Importantly, the data is showing that this is a very safe vaccine and folks are doing very well and actually having minimal, mild side effects.”
As previously noted, the Fort Meade MEDDAC hopes to get through the phase 1a population in the first two to three weeks. Salazar noted that the installation is following CDC (Centers for Disease Control) and Department of Defense prioritization policy. The next phase, phase 1b, includes personnel in critical national capability positions (phase 1b.1), personnel preparing to deploy overseas (phase 1b.2) and other critical support personnel not previously identified (phase 1b.3).
Phase 2 are high-risk beneficiaries and Phase 3 is the remaining healthy population. “We're highly encouraging everyone that when your time in the prioritization schema comes up to get the vaccination. I will be getting my vaccine for sure when it's my turn, and I just highly encourage everybody to take advantage of the protection this vaccine can offer,” said Salazar.
The questions remains for many, when do I get mine? After the initial Phase 1a vaccinations are completed the MEDDAC will have more information on availability and how long the process is taking.
Fort Meade MEDDAC and Kimbrough Ambulatory Care Center Commander, Colonel Tracy Michael will make his regular appearance on the next installation town hall, Thurs., Jan. 14, to take questions from the community and provide medical updates.